Phosphate esters and phosphoramides are important organophosphorus compounds, which have been widely used as functional molecules in the fields of ligands, organic synthesis, medicine, pesticides and flame retardants.However, the traditional synthesis methods of these compounds depend on the conversion of toxic phosphoryl halides or harsh reaction conditions without water and oxygen.The Atherton Todd reaction is one of the most effective methods for the preparation of phosphates. In this reaction, phosphoryl chloride generated by the interaction of P (o) H compounds with chlorination reagents reacts with alcohols to form phosphate (scheme 1a).Due to the low activity of phosphorus anion produced by P (o) H compounds under alkaline conditions, highly toxic carbon tetrachloride is usually required as chlorination reagent and solvent.Since then, a series of improvements have been made to the Atherton Todd reaction, but there are still some shortcomings in the existing methods. Therefore, it is still of great significance to develop simple and efficient synthesis methods for this kind of important organophosphorus compounds.In recent years, Associate Professor Chen Qian and Professor Huo Yanping of organic optoelectronic materials and functional polymers team of College of light industry and chemical engineering have carried out a series of phosphorylation reactions (2016, 81, 276; 2016, 81, 9476; 2016, 81, 10043; 2018, 360, 3590; 2018, 5, 2652; 2019, 84, 14949).On the basis of this research, the author further developed the scheme 1b initiated by phosphorus free radical in air atmosphere for the first time by drawing lessons from the strategy of our research group (2019, 21, 798), and the relevant research results were published online on (with the title of "" recently).J. Org. Chem.J. Org. Chem.J. Org. Chem.Adv. Synth. Catal.Org. Chem. Front.J. Org. Chem.Green Chem.Photoradio initiated Atherton – Todd reaction under open air chemical communications doi: 10.1039/c9cc09407e
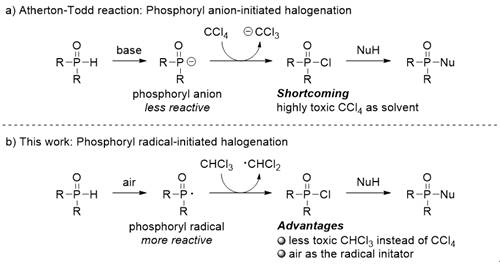
Scheme 1The Atherton–Todd-type reaction.
(source:)Chem. Commun.
In this reaction, air was used as the free radical initiator, and P (o) H compound produced phosphorus free radical by self oxidation in air.Then, the highly active p-radicals grab the chlorine atoms in chloroform to form phosphoryl chloride, and the dichloromethyl radical can further grab the protons in P (o) H compounds to produce new phosphorus radicals.Finally, phosphoryl chloride reacts with nucleophilic reagent to form phosphorylation product (scheme 2).Compared with the traditional Atherton−Todd reaction, this method has the following advantages: 1) replacing highly toxic carbon tetrachloride (reaction solvent) with 1 equivalent of chloroform with low toxicity; 2) using environment-friendly air as phosphorus free radical initiator; 3) various suitable reaction solvents; 4) wide substrate adaptability; 5) 10 g scale reaction.
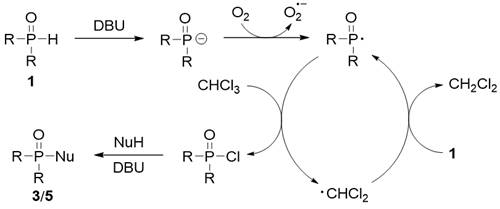
Scheme 2Proposed mechanism.
(source:)Chem. Commun.
Qu yingcong, a master's student of the Institute of light industry and chemical engineering, is the first author of the paperAssociate Professor Chen Qian is the corresponding author of the paper, and Guangdong University of technology is the first unit of the paper.The research was supported by "Guangdong science and technology program" (2017a010103044), "Guangdong Key Laboratory of functional molecular engineering open fund" (2016kf07) and "Guangdong University of Technology Youth hundred talents program" (220413506).
 current position:
Home
>
Research Progress
>
Content
current position:
Home
>
Research Progress
>
Content