Recently, the research group of Professor Lin Zhan of our college has made important progress in the field of battery recycling. The research results were published in nature communications (if = 12.353, the top journal in the first district), entitled "sustainability inspired cell design for a fully recyclable substance ion battery".Professor Lin Zhan is concerned about the sustainable development of the new energy industry, emphasizing that the large-scale production, use and disposal of secondary batteries should not be at the expense of the environment; human beings have experienced the crisis of "white pollution", and can not repeat the same mistakes in the utilization of secondary batteries.As the substitute of lithium-ion battery, sodium ion battery can be used in large-scale fixed energy storage. The recycling of waste sodium ion battery deserves serious consideration of researchers.Based on the structure design of sodium ion battery, the feasibility of battery recycling was considered. The bipolar structure was used in the sodium ion battery, which effectively promoted the rapid separation of battery components, and achieved almost 100% recovery of electrode materials.Dr. Liu Tiefeng is the first author of the paper, Professor Lin Zhan is the first corresponding author, and our university is the first unit of the paper.
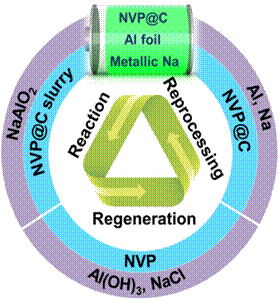
In the manufacturing process of lithium-ion battery, aluminum foil and copper foil are used as positive and negative collector respectively. This design is determined by the properties of battery materials, but it seriously affects the separation and purification efficiency of waste battery components.Sodium ion battery uses the structural components of lithium-ion battery. After the battery is scrapped, it is also faced with the problem of recycling and reprocessing.As the substitute of lithium-ion battery, the huge production and scale of sodium ion battery will inevitably cause pollution and damage to the environment.Therefore, before the manufacture of sodium ion battery, it is necessary to consider the recyclability of battery to reduce the difficulty of recycling waste sodium ion battery; however, it is difficult to re recognize and design the structure of secondary battery.
Professor Lin Zhan's research group adopts the bipolar battery structure, simplifies the composition of sodium ion battery, and designs a bipolar sodium ion battery based on the characteristics that aluminum foil and sodium do not produce alloying electrochemical reaction, so as to realize the efficient separation and recycling of electrode materials.Almost 100% of the electrode materials were recovered without adding new electrode materials, and the recovery rate of solid materials was 98.3%.In addition, the electrochemical properties of the recovered electrode materials recovered well after activation, and the electrode materials could be recycled repeatedly (as shown in the figure).Therefore, redesigning the battery structure and improving the recovery of electrode materials will play an important role in guiding the development and application of secondary batteries in the future.
Paper links:https://www.nature.com/articles/s41467-019-09933-0
Lin Zhan, distinguished professor and doctoral supervisor of School of light industry and chemical engineering, Guangdong University of technology, mainly engaged in the design and synthesis of new energy materials and devices and related basic research.So far, in Chem. Rev. (if = 52.613) energy environment. SCI. (if = 30.067), NAT. Commun. (if = 12.353), JAm.Chem. Soc.(IF=14.357), Angew. Chem. Int. Ed.(IF=12.102), Adv. energy mater. (if = 21.875) and other journals have published more than 70 academic papers. Among them, 20 papers have been published in journals with impact factor if > 10 as the first author and corresponding author. Three U.S. patents and three Chinese patents have been applied, and two chapters of works have been published. The research results have been cited more than 5000 times by peers in more than 30 countries and regions in the world, h-index = 33.
 current position:
Home
>
Research Progress
>
Content
current position:
Home
>
Research Progress
>
Content